BARCELONA, Spain – Patients with mesothelioma may gain similar benefit from immunotherapy as chemotherapy, and good responders may provide important clues to novel treatment for the thousands of new cases each year. Data from the PROMISE-meso trial presented at the ESMO Congress 2019 highlight the need to understand the biological mechanisms whereby mesothelioma, which is incurable, adapts to immunotherapy in some patients but not in others, resulting in variations in treatment response. (1)
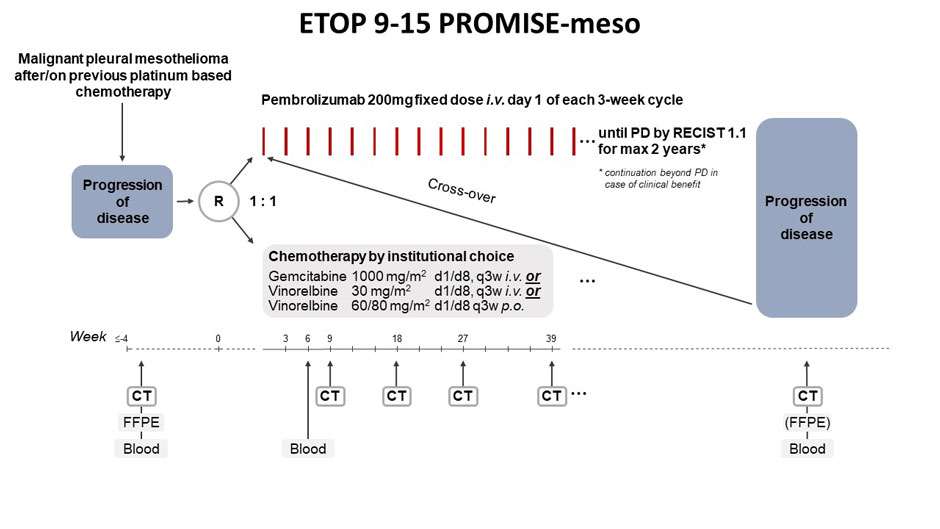
PROMISE-meso compared the effects of immunotherapy with the checkpoint inhibitor, pembrolizumab, and standard chemotherapy after patients with mesothelioma relapsed on or after first-line treatment.
“In PROMISE-meso, nearly four times more patients responded to immunotherapy than standard chemotherapy, but unfortunately these responses did not delay progression or improve survival. These findings are disappointing but, as in previous studies, some patients benefitted from immunotherapy for long periods. If we can find out how this happens, we will have a better idea of which patients should preferentially receive this treatment over chemotherapy,” said study author Dr Sanjay Popat, Royal Marsden Hospital NHS Foundation Trust, London, UK. “Nevertheless, whilst pembrolizumab was not superior to chemotherapy, survival was similar, and so pembrolizumab may represent an alternative.”
Popat explained that new checkpoint inhibitor studies are needed in patients with earlier stages of mesothelioma than those in PROMISE-meso. Studies also need to investigate the potential of combining immunotherapy with other anti-cancer drugs.
“In studies of lung cancer, we have already learned that we can improve results with immunotherapy by combining it with chemotherapy and the same may be true with mesothelioma. I would advise clinicians to enroll their patients into one of the large ongoing trials of first-line combination treatment so we can get answers as soon as possible about how to improve mesothelioma treatment,” said Popat. “Meanwhile, we need to better understand which patients benefit most from immunotherapy.”
Mesothelioma is a rare but fatal form of thoracic cancer that is diagnosed in more than 30,000 people per year and kills over 25,000. (2) Over 80% of cases arise from exposure to asbestos fibres which cause long term inflammation in the mesothelial cells of the lung, slowly leading to cancerous changes 20-50 years later. (3) The incidence of mesothelioma has fallen in Australia, the USA and Western Europe, where asbestos or strict regulations were introduced in the 1970s and 1980s. (4) Deaths in the USA have gradually decreased compared with Western Europe where mortality is relatively stable. (5) Deaths in Eastern Europe appear to be rising, possibly due to later asbestos bans, and rates are also rising in Japan due to historical asbestos imports. (5)
“The worldwide number of deaths is expected to rise as people exposed to asbestos before it was banned continue to be diagnosed many years later,” said Dr. Federica Grosso, Mesothelioma and Rare Cancers Unit, Azienda Ospedaliera SS Antonio e Biagio e Cesare Arrigo, Alessandria, Italy. “In some ‘hotspots’ such as Casale Monferrato in Italy which had the largest asbestos plant in the world until it was closed in 1987, mesothelioma diagnosis and death are common. In a population of 35,000, there are approximately 50 cases per year – an incidence more than 20 times higher than in the rest of the country.”
“Mesothelioma is a huge problem because asbestos powder from the plants pollutes large areas. It isn’t just people who worked in the plant who are being diagnosed, it is their families and unrelated people, some of whom are only 40-50 years old – much younger than we would expect to see with mesothelioma,” she pointed out.
“A similar situation of environmental exposure was recently reported in Sibatè (Colombia) where a plant was closed only a few years ago and many cases of mesothelioma are being diagnosed. Unfortunately, we can expect to see an increase in mesothelioma in countries where asbestos is still used for many years to come,” she added. “Asbestos has been totally or partially banned in only 66 nations worldwide, most of them high-income countries, and most of the current consumption and production is concentrated in lower and middle income countries.”
Grosso explained that treatment of mesothelioma is limited, with the only approved regimen being the combination of pemetrexed and platinum derivatives. Patients generally die within two years of diagnosis. (4) There is currently no standard effective second line therapy for patients with mesothelioma. Those who respond well to pemetrexed and platinum based chemotherapy may be given a repeat course while others are usually offered drugs such as gemcitabine/vinorelbine with response rates of approximately 10%.
“Although we did not see better survival with immunotherapy in the PROMISE-meso study, the responses are encouraging and the results of the ongoing trials of checkpoint inhibitor treatment in earlier stage mesothelioma will be very important to patients and clinicians. We very much need better first and second-line treatment for mesothelioma around the world,” concluded Grosso.
Study results
In the phase III PROMISE-meso trial (NCT02991482), 144 patients with advanced pre-treated mesothelioma were randomised to pembrolizumab (200 mg every three weeks) or the participating centre’s choice of standard chemotherapy (gemcitabine/vinorelbine). Patients in the control group were able to cross over to pembrolizumab at progression.
The objective response rate (ORR) was 22% in patients treated with pembrolizumab compared to 6% with chemotherapy (p=0.004). Median progression-free survival (PFS) was 2.5 months (95% CI 2.1-4.2) and 3.4 months (2.2-4.3) respectively (p=0.76). Median overall survival was 10.7 months for pembrolizumab vs 11.7 months for chemotherapy (p=0.85). Treatment-related adverse events grade ≥3 were experienced by 19% of patients in the pembrolizumab group and 24% in the chemotherapy group, one fatal in each group.
Official Congress Hashtag: #ESMO19
Social Media information
References
- LBA91_PR ‘A multicenter randomized Phase III trial comparing pembrolizumab versus standard chemotherapy for advanced pre-treated malignant pleural mesothelioma (MPM) – results from the European Thoracic Oncology Platform (ETOP 9-15) PROMISE-MESO trial’ will be presented by Sanjay Popat during the Proffered paper session on Monday 30 September, 10:15-11:45 CEST in Pamplona Auditorium (Hall 2). Annals of Oncology, Volume 30, Supplement 5, October 2019
- Bray F, Ferlay J, Soerjomataram I et al. Global Cancer Statistics 2018: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin 2018; 68: 394-424
- Editorial. Asbestos exposure: the dust cloud lingers. Lancet Oncology 2019; 20:1035
- Carbone M, Adusumilli PS, Alexander HR Jr et al. Mesothelioma: Scientific clues for prevention, diagnosis, and therapy. CA Cancer J Clin. 2019 Sep;69(5):402-429.
- Boffetta P, Malvezzi M, Pira E et al. International Analysis of Age-Specific Mortality Rates From Mesothelioma on the Basis of the International Classification of Diseases, 10th Revision. Glob Oncol. 2018; 4: JGO.2017.010116.
- British Lung Foundation. What is asbestos?
About asbestos and mesothelioma (6)
Asbestos is a term for a group of minerals that were widely mined during the 20th century for use in buildings for insulation, flooring and roofing, and sprayed on ceilings and walls. Asbestos is made up of microscopic fibres which may be released into the air when asbestos is broken up. If fibres are inhaled, they can damage the two-layered membrane around the lungs and lining the rib cage called the pleura. Following exposure to asbestos, pleural cells may slowly become cancerous, either forming one large tumour or many small tumours scattered through the pleural membrane. This is mesothelioma.
People who work in the building and construction industries, including carpenters, electricians, plumbers, sheet metal workers, painters and decorators, are at increased risk of exposure to asbestos and mesothelioma. Family members are also at risk as they may inhale asbestos fibres off clothing of those working with asbestos.
Symptoms of mesothelioma include breathlessness, coughing and chest pain. Although mesothelioma takes many years to appear after exposure to asbestos, it then progresses rapidly.
LBA91_PR - A multicenter randomized phase III trial comparing pembrolizumab versus standard chemotherapy for advanced pre-treated malignant pleural mesothelioma (MPM) – results from the European Thoracic Oncology Platform (ETOP 9-15) PROMISE-meso trial
S. Popat1, A. Curioni-Fontecedro2, V. Polydoropoulou3, R. Shah4, M. O'Brien5, A. Pope6, P. Fisher7, J. Spicer8, A. Roy9, D. Gilligan10, O. Gautschi11, E. Nadal12, W.-D. Janthur13, R. López Castro14, R. García Campelo15, H. Roschitzki-Voser16, B. Ruepp16, S. Rusakiewicz17, S. Peters18, R.A. Stahel19
1Medicine, Royal Marsden Hospital NHS Foundation Trust, London, United Kingdom, 2Center Of Hematology And Oncology, University Hospital, Zürich, Switzerland, 3Frontier Science Foundation-Hellas, Frontier Science Foundation-Hellas, Athens, Greece, 4Oncology, Kent Oncology Centre, Maidstone, United Kingdom, 5Medicine, Royal Marsden Hospital Sutton, London, United Kingdom, 6Oncology, Clatterbridge Cancer Centre, Liverpool, United Kingdom, 7Oncology, Weston Park Hospital, Sheffield, United Kingdom, 8Comprehensive Cancer Centre, King's College London Guy's Hospital, London, United Kingdom, 9Oncology, Plymouth Hospitals NHS Trust, Plymouths, United Kingdom, 10Oncology, Addenbrooke's Hospital, Cambridge, United Kingdom, 11Oncology, Cantonal Hospital Luzern and Swiss Group for Clinical Cancer Research, Luzern, Switzerland, 12Oncology, Institut Catala d'Oncologia (ICO), Barcelona, Spain, 13Oncology, Cantonal Hospital Aarau and Swiss Group for Clinical Cancer Research, Aarau, Switzerland, 14Department Of Oncology, Hospital Clínico Universitario de Valladolid, Valladolid, Spain, 15Oncology, Hospital Teresa Herrera, La Coruña, Spain, 16Coordinating Office, European Thoracic Oncology Platform (ETOP), Bern, Switzerland, 17Centre Of Experimental Therapies (cte), Department Of Oncology, Centre Hospitalier Universitaire Vaudois (CHUV), Lausanne, Switzerland, 18Department Of Oncology, Centre Hospitalier Universitaire Vaudois, Lausanne, Switzerland, 19Cccz, University Hospital Zürich, Zurich, Switzerland
Background: MPM is an aggressive malignancy of increasing prevalence and poor prognosis. At relapse after platinum-based (pb) CT, single agent CT is commonly used and single arm trials of immune checkpoint inhibitors have demonstrated encouraging activity.
Methods: PROMISE-meso is an open-label 1:1 randomized phase III trial investigating the efficacy of P (200 mg/Q3W) vs institutional choice single agent CT (gemcitabine or vinorelbine) in relapsed MPM patients (pts) failing one previous line of pb CT. Pts were of PS 0-1 and unselected for PD-L1 status. P beyond progression (PD) for clinical benefit and crossover to P at PD on CT were allowed. Primary endpoint was progression-free survival (PFS, RECIST 1.1) by independent radiological review (IR). The trial was designed to detect an increase in median PFS from 3.5 months (ms) to 6ms with P (HR=0.58, 80% power, 1-sided α=0.025). 142 pts were needed to observe the required 110 events. Secondary endpoints were overall survival (OS), investigator assessed (IA) PFS, objective response rate (ORR), adverse events (AE), while efficacy by PD-L1 status was exploratory.
Results: Between 09/17 and 08/18, 144 pts were randomized, 73 to P and 71 to CT. At 20/02/19 data lock, 70 pts were on follow-up (median 12ms). Pts were of median age 70 years, 82% males, 77% poor EORTC prognostic score, 50% never smokers, 89% epithelioid histology and 65% (of 102 available) TPS≥1%. ORRs were 22% in P, 6% in CT (p=0.004). 62 IR PFS events were observed in P vs 56 in CT, median PFS 2.5ms (95%CI 2.1-4.2) vs 3.4ms (2.2-4.3), HR=1.06 (0.73–1.53), p=0.76. Median OS was 10.7ms for P vs 11.7ms for CT, HR=1.05 (0.66-1.67), p=0.85. 45 CT pts crossed over to P. Accounting for crossover yielded similar OS results. Treatment-related AEs grade ≥3 were experienced by 19% P vs 24% CT pts, one fatal per arm. Most common AEs were fatigue (19%) in P vs nausea (27%) and fatigue (31%) in CT.
Conclusions: This is the first randomized trial evaluating the efficacy of P vs single agent CT in MPM pts progressing after or on previous pb CT. In unselected pts, whilst associated with an improved ORR, P does not improve PFS or OS over single agent CT.
Clinical trial identification: NCT02991482
Legal entity responsible for the study: European Thoracic Oncology Platform (ETOP)
Funding: MSD Merck Sharp & Dohme AG
Disclosure: S. Popat: Honoraria (self), Advisory / Consultancy, Research grant / Funding (institution), Travel / Accommodation / Expenses: Boehringer Ingelheim; Research grant / Funding (institution): Epizyme; Advisory / Consultancy, Research grant / Funding (institution), Travel / Accommodation / Expenses: BMS; Research grant / Funding (institution): Clovis Oncology; Honoraria (self), Advisory / Consultancy, Research grant / Funding (institution): Roche; Research grant / Funding (institution): Lilly; Honoraria (self), Research grant / Funding (institution): Takeda; Honoraria (self), Advisory / Consultancy: AstraZeneca; Honoraria (self): Chugai Pharma; Advisory / Consultancy: Novartis; Advisory / Consultancy: Pfizer; Advisory / Consultancy, Travel / Accommodation / Expenses: Merck Sharp & Dohme; Advisory / Consultancy: Guardant Health; Advisory / Consultancy: Abbvie.
A. Curioni-Fontecedro: Honoraria (self), Advisory / Consultancy: AstraZeneca; Honoraria (self), Advisory / Consultancy: Boehringer Ingelheim; Honoraria (self), Advisory / Consultancy: Bristol-Myers Squibb; Honoraria (self), Advisory / Consultancy: Roche; Honoraria (self), Advisory / Consultancy: Merck Sharp and Dohme; Honoraria (self), Advisory / Consultancy: Novartis; Honoraria (self), Advisory / Consultancy: Pfizer; Honoraria (self), Advisory / Consultancy: Takeda.
R. Shah: Advisory / Consultancy: Merck Sharp & Dohme; Advisory / Consultancy: Lilly.
M. O'Brien: Advisory / Consultancy: Merck Sharp & Dohme; Advisory / Consultancy: BMS; Advisory / Consultancy: Abbvie; Advisory / Consultancy: Pierre fabre; Advisory / Consultancy: Boehringer Ingelheim; Advisory / Consultancy, Teaching role for Roche: Roche.
P. Fisher: Advisory / Consultancy, Travel / Accommodation / Expenses: Merck Sharp & Dohme.
D. Gilligan: Honoraria (self): Merck Sharp & Dohme.
E. Nadal: Advisory / Consultancy: Merck Sharp & Dohme.
R. López Castro: Honoraria (self), Travel / Accommodation / Expenses: Takeda; Honoraria (self), Travel / Accommodation / Expenses: AstraZeneca; Honoraria (self), Advisory / Consultancy, Research grant / Funding (self), Travel / Accommodation / Expenses: Boehringer Ingelheim; Honoraria (self), Research grant / Funding (self), Travel / Accommodation / Expenses: Bristol-Myers Squibb; Honoraria (self), Travel / Accommodation / Expenses: Novartis; Honoraria (self), Advisory / Consultancy, Research grant / Funding (self), Travel / Accommodation / Expenses: Roche; Honoraria (self), Travel / Accommodation / Expenses: Merck Serono; Honoraria (self), Travel / Accommodation / Expenses: Pfizer; Advisory / Consultancy: Aristo.
R. García Campelo: Advisory / Consultancy, Speaker Bureau / Expert testimony: Merck Sharp & Dohme.
S. Peters: Honoraria (self): Abbvie; Honoraria (self): Amgen; Honoraria (self): AstraZeneca; Honoraria (self): Bayer; Honoraria (self): Biocartis; Honoraria (self): Boehringer-Ingelheim; Honoraria (self): Bristol-Myers Squibb; Honoraria (self): Clovis; Honoraria (self): Daiichi Sankyo; Honoraria (self): Debiopharm; Honoraria (self): Lilly; Honoraria (self): Roche; Honoraria (self): Foundation Medicine; Honoraria (self): Illumina; Honoraria (self): Janssen; Honoraria (self): Merck Sharp and Dohme; Honoraria (self): Merck Serono; Honoraria (self): Merrimack; Honoraria (self): Novartis; Honoraria (self): Pharma Mar; Honoraria (self): Pfizer; Honoraria (self): Regeneron; Honoraria (self): Sanofi; Honoraria (self): Seattle Genetics ; Honoraria (self): Takeda.
R.A. Stahel: Honoraria (self): Abbvie; Honoraria (self), Research grant / Funding (self): AstraZeneca; Honoraria (self), Research grant / Funding (self): Boehringer Ingelheim; Honoraria (self), Research grant / Funding (self): Merck Sharp & Dohme; Honoraria (self), Research grant / Funding (self): Pfizer; Honoraria (self), Research grant / Funding (self): Roche; Honoraria (self): Takeda; Research grant / Funding (self): BMS; Research grant / Funding (self): Genentech.
This press release contains information provided by the author of the highlighted abstract and reflects the content of this abstract. It does not necessarily reflect the views or opinions of ESMO who cannot be held responsible for the accuracy of the data. Commentators quoted in the press release are required to comply with the ESMO Declaration of Interests policy and the ESMO Code of Conduct.